Duchenne Muscular Dystrophy (DMD) Market Analysis, Market Size, Epidemiology, Leading Companies
- Ankit Nigam
- May 8, 2020
- 4 min read
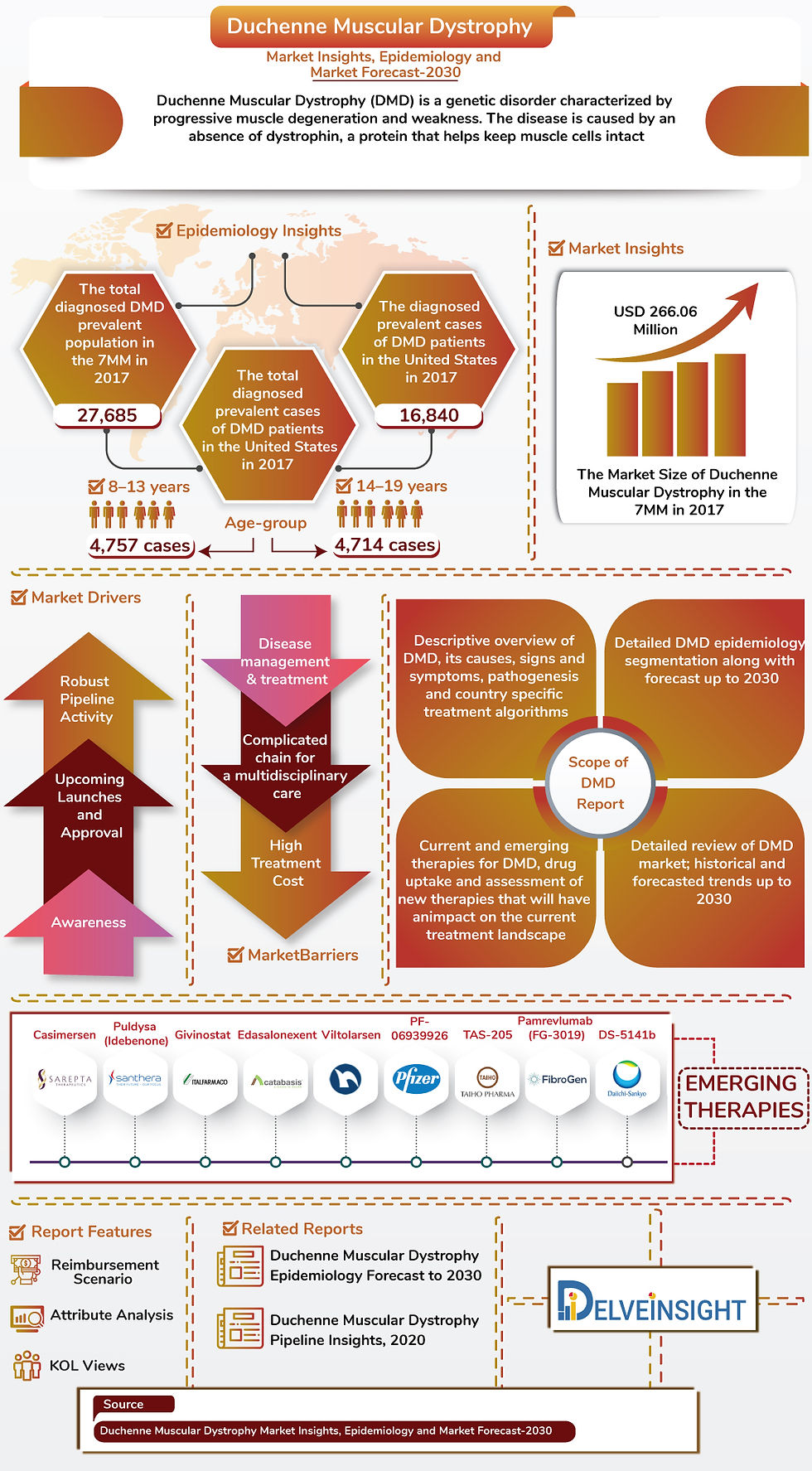
The growth of Duchenne Muscular Dystrophy (DMD) Market is anticipated to increase because of an increasing incidence of DMD patients as well as the launch of emerging drugs in the 7MM.
DelveInsight has introduced a new Market Insights, Epidemiology, and Market Forecast Report on "Duchenne Muscular Dystrophy Market Insights, Epidemiology and Market Forecast-2030" to its portfolio.
Key Highlights from the report are:
As per the DelveInsight analysis, the total diagnosed Duchenne Muscular Dystrophy prevalent population in the 7 MM was 27,685 in 2017.
The total diagnosed prevalent cases of DMD patients were found to be maximum in the age-group of 8–13 year and 14–19 year in the United States in 2017.
The treatment for DMD patients is the standard care along with the new upcoming therapeutic strategies covering Genetic Therapies, Cell therapy using muscle precursor cells or stem cells, Membrane stabilization and upregulation of cytoskeletal proteins and secondary cascades treatment.
Duchenne Muscular Dystrophy market size in the 7MM is expected to increase during the study period. As per the estimates, the highest contribution in the market size of DMD is from the United States, followed by EU-5 countries and Japan.
Duchenne Muscular Dystrophy is a progressive form of muscular dystrophy, which usually occurs in males, though in rare cases may affect females as well, causing progressive weakness and loss (atrophy) of skeletal and heart muscles.
The disease epidemiology covered in the report proffers historical as well as forecasted Duchenne Muscular Dystrophy epidemiology, which is segmented as Diagnosed Prevalent Population of DMD, Age-specific Diagnosed Prevalence of DMD, Mutation-specific Diagnosed Prevalence of DMD and Diagnosed Prevalence of Associated Comorbidities in DMD in the 7MM market from 2017 to 2030.
The report also covers Mutation-specific Diagnosed Prevalence of Duchenne Muscular Dystrophy, including several mutations such as Large Mutations, Small Mutations and Point Mutations with major proportion for deletions in Large Mutations subgroup.
Duchenne Muscular Dystrophy report encloses the detailed analysis of DMD marketed drugs and mid and late-stage pipeline drugs.
The therapies that are approved for the DMD treatment are Vyondys 53 (Golodirsen), Emflaza, Exondys 51, Translarna along with many more.
Drugs covered in the report are:-
There are several key players robustly involved in developing potential products such as
Casimersen
SRP-9001
Puldysa (Idebenone)
Givinostat
Edasalonexent
Viltolarsen
PF-06939926
Vamorolone
TAS-205
Pamrevlumab (FG-3019)
Allogeneic Cardiosphere-Derived Cells (CAP-1002)
DS-5141b
And many others
Key Players covered in the DMD market report are:-
Sarepta Therapeutics
Italfarmaco
Catabasis Pharmaceuticals
Nippon Shinyaku (NS Pharma)
Pfizer
Santhera Pharmaceuticals/ReveraGen BioPharma
Taiho Pharmaceutical
FibroGen
Capricor
Daiichi Sankyo
And many others
The reasons for buying this report:
The report proffers an overview of pathophysiology, various diagnostic approaches and Duchenne Muscular Dystrophy treatment algorithm, including detailed chapters for marketed products and emerging therapies.
Historical and forecasted DMD epidemiology in 7MM covering the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan from 2017-2030.
Detailed historical and forecasted Duchenne Muscular Dystrophy market covering the United States, EU5 and Japan from 2017-2030.
Pipeline analysis across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with comprehensive clinical profiles, key cross-competition, launch date along with product development activities.
Detailed DMD market size by therapies, covering the United States, EU5 and Japan from 2017-2030.
Reimbursement scenario and Key Opinion Leader Views.
Table of Contents
1. Key Insights
2. Executive Summary of Duchenne Muscular Dystrophy
3. Competitive Intelligence Analysis for Duchenne Muscular Dystrophy
4. Duchenne Muscular Dystrophy: Market Overview at a Glance
4.1. Duchenne Muscular Dystrophy Total Market Share (%) Distribution in 2017
4.2. Duchenne Muscular Dystrophy Total Market Share (%) Distribution in 2030
5. Duchenne Muscular Dystrophy: Disease Background and Overview
5.1. Introduction
5.2. Sign and Symptoms
5.3. Pathophysiology
5.4. Risk Factors
5.5. Diagnosis
6. Patient Journey
7. Duchenne Muscular Dystrophy Epidemiology and Patient Population
7.1. Epidemiology Key Findings
7.2. Assumptions and Rationale: 7MM
7.3. Epidemiology Scenario: 7MM
7.3.1. Duchenne Muscular Dystrophy Epidemiology Scenario in the 7MM (2017-2030)
7.4. United States Epidemiology
7.4.1. Duchenne Muscular Dystrophy Epidemiology Scenario in the United States (2017-2030)
7.5. EU-5 Country-wise Epidemiology
7.5.1. Germany Epidemiology
7.5.1.1. Duchenne Muscular Dystrophy Epidemiology Scenario in Germany (2017-2030)
7.5.2. France Epidemiology
7.5.2.1. Duchenne Muscular Dystrophy Epidemiology Scenario in France (2017-2030)
7.5.3. Italy Epidemiology
7.5.3.1. Duchenne Muscular Dystrophy Epidemiology Scenario in Italy (2017-2030)
7.5.4. Spain Epidemiology
7.5.4.1. Duchenne Muscular Dystrophy Epidemiology Scenario in Spain (2017-2030)
7.5.5. United Kingdom Epidemiology
7.5.5.1. Duchenne Muscular Dystrophy Epidemiology Scenario in the United Kingdom (2017-2030)
7.5.6. Japan Epidemiology
7.5.6.1. Duchenne Muscular Dystrophy Epidemiology Scenario in Japan (2017-2030)
8. Treatment Algorithm, Current Treatment, and Medical Practices
8.1. Duchenne Muscular Dystrophy Treatment and Management
8.2. Duchenne Muscular Dystrophy Treatment Algorithm
9. Unmet Needs
10. Key Endpoints of Duchenne Muscular Dystrophy Treatment
11. Marketed Products
11.1. List of Marketed Products in the 7MM
11.2. Drug Name: Company Name
11.2.1. Product Description
11.2.2. Regulatory Milestones
11.2.3. Other Developmental Activities
11.2.4. Pivotal Clinical Trials
11.2.5. Summary of Pivotal Clinical Trial
List to be continued in report
12. Emerging Therapies
12.1. Key Cross
12.2. Drug Name: Company Name
12.2.1. Product Description
12.2.2. Other Developmental Activities
12.2.3. Clinical Development
12.2.4. Safety and Efficacy
12.2.5. Product Profile
List to be continued in report
13. Duchenne Muscular Dystrophy: Seven Major Market Analysis
13.1. Key Findings
13.2. Duchenne Muscular Dystrophy Market Size in 7MM
13.3. Duchenne Muscular Dystrophy Market Size by Therapies in the 7MM
14. Attribute analysis
15. 7MM: Market Outlook
15.1. United States: Market Size
15.1.1. Duchenne Muscular Dystrophy Total Market Size in the United States
15.1.2. Duchenne Muscular Dystrophy Market Size by Therapies in the United States
15.2. EU-5 countries: Market Size and Outlook
15.3. Germany Market Size
15.3.1. Duchenne Muscular Dystrophy Total Market Size in Germany
15.3.2. Duchenne Muscular Dystrophy Market Size by Therapies in Germany
15.4. France Market Size
15.4.1. Duchenne Muscular Dystrophy Total Market Size in France
15.4.2. Duchenne Muscular Dystrophy Market Size by Therapies in France
15.5. Italy Market Size
15.5.1. Duchenne Muscular Dystrophy Total Market Size in Italy
15.5.2. Duchenne Muscular Dystrophy Market Size by Therapies in Italy
15.6. Spain Market Size
15.6.1. Duchenne Muscular Dystrophy Total Market Size in Spain
15.6.2. Duchenne Muscular Dystrophy Market Size by Therapies in Spain
15.7. United Kingdom Market Size
15.7.1. Duchenne Muscular Dystrophy Total Market Size in the United Kingdom
15.7.2. Duchenne Muscular Dystrophy Market Size by Therapies in the United Kingdom
15.8. Japan Market Outlook
15.8.1. Japan Market Size
15.8.2. Duchenne Muscular Dystrophy Total Market Size in Japan
15.8.3. Duchenne Muscular Dystrophy Market Size by Therapies in Japan
16. Access and Reimbursement Overview of Duchenne Muscular Dystrophy
17. KOL Views
18. Market Drivers
19. Market Barriers
20. Appendix
20.1. Bibliography
20.2. Report Methodology
21. DelveInsight Capabilities
22. Disclaimer
23. About DelveInsight
About DelveInsight
DelveInsight is a leading Business Consultant, and Market Research Firm focused exclusively on life sciences. It supports pharma companies by providing end to end comprehensive solutions to improve their performance.
Related Reports:-
Duchenne Muscular Dystrophy Epidemiology Forecast to 2030 DelveInsight's Duchenne Muscular Dystrophy - Epidemiology Forecast 2030 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Duchenne Muscular Dystrophy in the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan.
Duchenne Muscular Dystrophy Pipeline Insights, 2020 Duchenne Muscular Dystrophy Pipeline Insight, 2020 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the Duchenne Muscular Dystrophy market.


Comments